Our Science
 AlgonotTM products are based on many years of research supported by numerous scientific publications, clinical studies, patents, and trademarks by our Scientific Director, Theoharis C. Theoharides, BA, MS, MPhil, PhD, MD, Professor and Vice Chair, Clinical Immunology and Director, Institute of Neuro-immune Medicine-Clearwater, Nova Southeastern University, FL and Adjunct Professor, Tufts University School of Medicine, Boston, MA. Dr. Theoharides has 500 publications placing him in the world’s top 2% of most-cited authors and the top-rated expert worldwide on mast cells by Expertscape. He was inducted into the Alpha Omega Alpha National Medical Honor Society, the Rare Diseases Hall of Fame, and the World Academy of Sciences.
AlgonotTM products are based on many years of research supported by numerous scientific publications, clinical studies, patents, and trademarks by our Scientific Director, Theoharis C. Theoharides, BA, MS, MPhil, PhD, MD, Professor and Vice Chair, Clinical Immunology and Director, Institute of Neuro-immune Medicine-Clearwater, Nova Southeastern University, FL and Adjunct Professor, Tufts University School of Medicine, Boston, MA. Dr. Theoharides has 500 publications placing him in the world’s top 2% of most-cited authors and the top-rated expert worldwide on mast cells by Expertscape. He was inducted into the Alpha Omega Alpha National Medical Honor Society, the Rare Diseases Hall of Fame, and the World Academy of Sciences.The only liposomal luteolin supplements in olive pomace oil
Our Research
The Role of Mast Cells in Allergic and Neuroinflammatory Processes
Many individuals are sensitive to multiple environmental and other stimuli that can affect numerous tissues/organs and lead to a variety of symptoms, often termed “mystery symptoms” that can be very confusing. A key immune cell involved is the mast cell, that “orchestrates” the immune response of the body to a real or perceived danger. Activation of mast cells is typically associated with allergic reactions, but is also mandatory for inflammatory processes
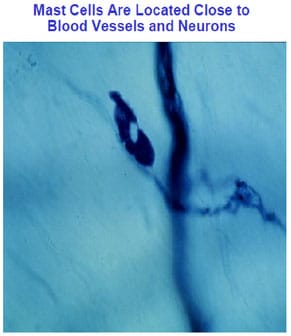
Mast cells are located close to blood vessels and neurons (Figure 1) in all tissues, especially those exposed to the outside world (eyes, nose, mouth, lungs, skin and gastrointestinal tract), but also the brain (see discussion later) Mast cells “mature” under the influence of local micro-environmental factors and can assume different phenotypes (mediators produced and reactivity) and have typically been divided based on their secretory granule content of proteolytic enzyme (proteases). Mucosal mast cells (MMC) contain only the chymase while connective tissue mast cells (CTMC) contain both chymase and tryptase. Histamine is stored in the secretory granules of both subtypes. Mast cell granules also store tissue remodeling enzymes, such as carboxypeptidase A3 (CPA3) and matrix metalloproteinases (MMPs). However, mast cell phenotypes may be modified depending on the triggers.
Pathophysiology of mast cells.
Mast cells are known to be triggered by immunoglobulin ( (IgE) binding to specific receptors FcεRI on their cell surface (Figure 2), but also multiple other surface receptors for a variety of stimuli, including positively charged molecules and drugs (MRGPRX2), for peptides released from nerves such as corticotropin-releasing hormone (CRH), hemokinin-1 (HK-1), nerve growth factor (NGG), neurotensin (NT) and substance P (SP), as well as receptors for immune molecules such as interleukins (IL-1β, IL-33) and for pathogens, including bacteria, fungi and viruses (toll-like receptors, TLRs).
When mast cells are triggered by FcεRI or via MRGPRX2, there is rapid secretion (degranulation shown on the left hand-side colored photomicrograph and the left-hand side bottom center electron photomicrograph) of many preformed molecules, stored in as many as 1000 secretory granules per cell, some of which are shown in the box on the lower right-hand side of Figure 1. Histamine and tryptase are the best known prestored molecules, but leukotrienes (involved in bronchoconstriction) and prostaglandins (involved in pain) are also rapidly produced during mast cell stimulation. Histamine is also released from basophils, which circulate in the blood. Histamine, leukotrienes and prostaglandins are rapidly degraded and are seldom measured in the blood; instead, their breakdown products (metabolites) are measured in 24-hour urine that has to be kept cold.
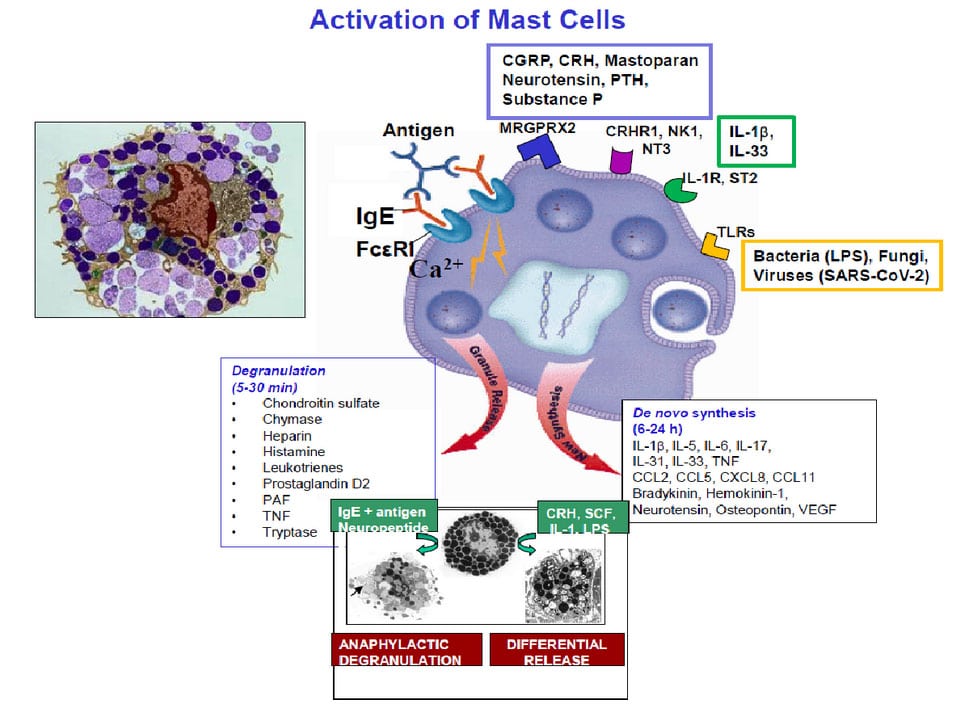
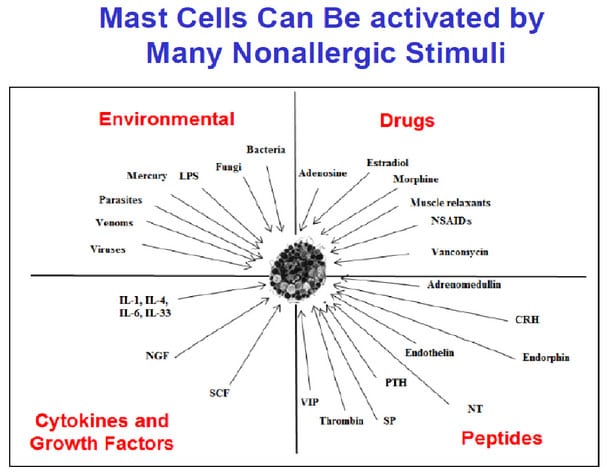
In addition to allergic stimulation, mast cells are stimulated by numerous non-allergic triggers (Figure 3), especially via activation of neuropeptide, interleukin or TLR receptors, they synthesize new inflammatory mediators (some of which are shown in the box on the lower right-hand side of Figure 1). that are released (differential release) without histamine and without degranulation (right-hand side bottom center electron photomicrograph). Tryptase has been studied extensively because it is considered an exclusive mast cell-associated mediator and is released during anaphylaxis and systemic mastocytosis as well as in some cases of mast cell activation. Molecules such IL-6 and vascular endothelial growth factor (VEGF) may reflect non-allergic stimulation, edema and inflammation. By rapidly releasing these mediators, mast cells actively interact with other cell types in their surrounding environment and participate in the induction and/or propagation of various allergic, inflammatory and immune responses.
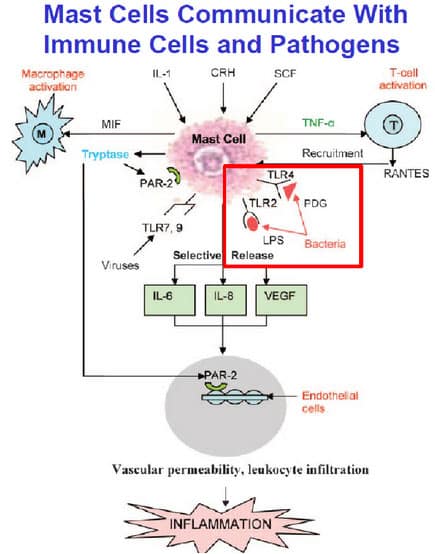
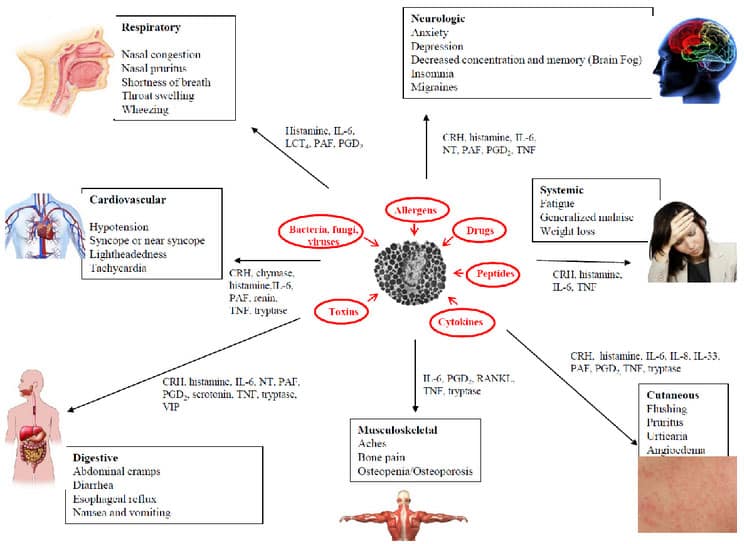
Mast cells also communicate with other immune cells such as macrophages (M) and T-cells (T) as shown in Figure 4. Once activated, mast cells secrete molecules such as IL-6, IL-8 and TNF, which lead to inflammation (Figure 4). Depending on the trigger and the different mediators released, mast cells can affect any and all organs of the body. Including the brain, leading to a very confusing, multisystemic symptomatology (Figure 5). In fact, affected individuals can present with symptoms mimicking many different overlapping diagnoses.
Mast cells in the Central Nervous System (CNS).
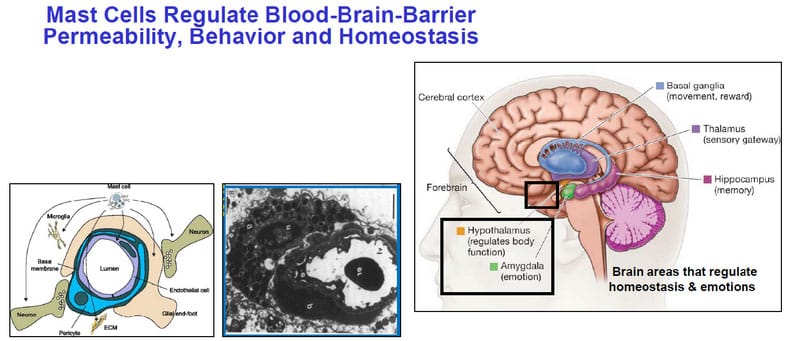
Mast cells are also present in the CNS, especially in the meninges, thalamus and hypothalamus, which regulate homeostasis. Mast cells are the richest source of histamine in the CNS. Brain mast cells had been termed the “immune gate to the brain” (Figure 6) since they “hug” the protective blood-brain-barrier (BBB). Mast cell-derived vasodilatory mediators (bradykinin, histamine, tryptase, VEGF) can disrupt the BBB, activate the brain’s defenders, the microglia, leading to localized inflammation, 006Fidative stress, disrupted neuronal communication, neurodegeneration and cognitive dysfunction, commonly known as “brain fog.”
Microglia in Neuroinflammation.
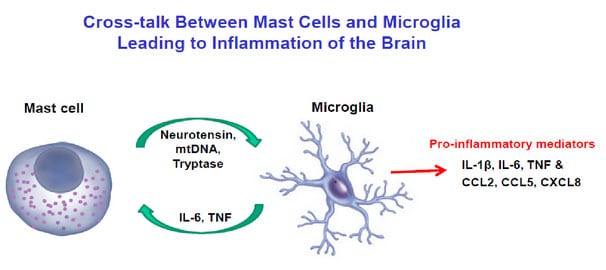
Microglia have been considered key players in brain injury, as well as the development of neuroinflammation and neurodegeneration. Upon stimulation, activated microglia adopt an ameboid shape that renders them capable of phagocytosis and release of pro-inflammatory cytokines and free radicals. Mast cells communicate with microglia (Figure 7) leading to their activation and contributing to neuroinflammation. Activation of mast cells and microglia, especially in the hypothalamus and the hippocampus, could lead to cognitive dysfunction and neuronal death. The effect of stress on mast cells has been well documented to both stimulate mast cells directly without secretion of histamine and tryptase, but also increase the reactivity of mast cells to many other triggers. Such an increase in mast cell reactivity has been reported after major surgery, trauma or other severe stressor. There are still no clinically effective inhibitors of mast cell activation (“mast cell stabilizers”). Most treatment approaches use anti-histamines or anti-leukotrienes. Biologics have been used to neutralize the effect of IgE or cytokines such as IL-4 and IL-13, but they do not inhibit activation of mast cells.
Minimizing exposure to potential triggers (e.g. allergens, foods, heat, stress) is clearly important, especially avoiding histamine-rich foods (e.g. avocado, cheese, eggplant, nectarines, sardines, spinach, spices, tomatoes). Supplementation with the main histamine metabolizing enzyme, diamine oxidase (DAO) shortly before meals could also be beneficial.
Certain naturally occurring flavonoids, commonly found in fruits, green plants and seeds, have potent antioxidant and anti-inflammatory properties. However, not all of them are safe and many (polyphenolics like resveratrol) can cause hyperactivity, while others (like soy flavonoids) could be detrimental because of their estrogenic effects. Moreover, flavonoids are difficult to dissolve and less than 10% are absorbed from the intestine in powder form thus leading to flavonoid accumulation in the gut and inhibition of gut enzymes causing dysbiosis. To make matters worse, most supplements do not indicate either the source or the purity of the flavonoids (e.g. a cheap source is peanut shells) with the risk of unexpected adverse reactions.
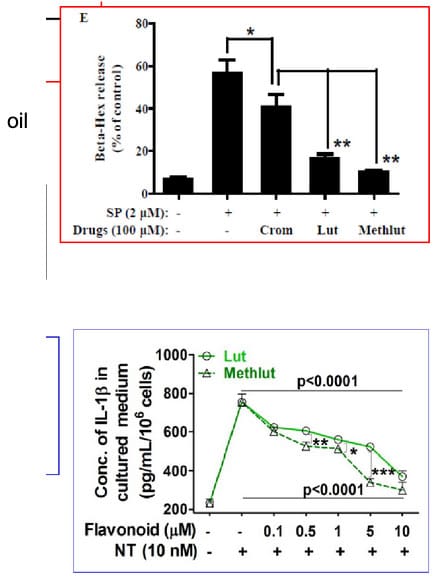
After 30 years of studies with numerous publications and patents, we concluded that the flavone luteolin (Lut) -not lutein which is a carotenoid- stands out because it significantly inhibits the activation of both mast cells and microglia (Figure 8). We then showed that tetramethoxyluteolin (Methlut), the novel structural analogue of luteolin, is an even more potent inhibitor than luteolin (figure 8) and has been incorporated into a skin lotion (GentleDerm®) because unlike other flavonoids, it has no color.
AlgonotTM has formulated luteolin with or without quercetin (98% purity, assayed by the independent laboratory Urofins) in olive pomace oil (softgel capsules) to increase intestinal absorption and provide the added benefits of unsaturated fatty acids and anti-inflammatory actions of olive oil.
Cognizant of the fact that some 20% of individuals are sensitive to phenolic compounds (found in berries, flavonoids and chocolate), AlgonotTM created the trademarked and patent-protected PureLut® (only luteolin), NeuroProtek-Low Phenol® (same amount of luteolin but less quercetin) and NeuroProtek-Low Phenol-Liquid® in dropper bottles for children who cannot swallow capsules.
As a way of helping the unsuspected consumers, we published an editorial where most available dietary supplements containing luteolin were compared (Biofactors). It was obvious that the labels were very misleading because purity and source were not disclosed, in some instances the content had no color (luteolin and quercetin are yellow), while in other instances small print indicated you had to take multiple doses to reach the amount stated on the label. In any event, combination of flavonoids should not exceed 2,000 mg/day without medical supervision as they may inhibit liver metabolic enzymes.







