FibroProtek®
FibroProtek® is a patented, all-natural dietary supplement in softgel capsules aimed at reducing widespread body pain, inflammation, allergy-related symptoms, and fatigue. It features a unique blend of pure flavonoids, based on rigorous scientific research by Dr. Theoharides and his colleagues. Using olive pomace oil for better absorption, FibroProtek® offers the benefits of Mediterranean olives. It is free from artificial additives and common allergens, including gluten, dairy, soy, and preservatives.
$49.00
3027 in stock (can be backordered)
Product Description
FibroProtek® is a unique, patented, all-natural oral dietary supplement formulated in softgel capsules, which may reduce widespread body pain, inflammation, allergy related symptoms and fatigue. It features a unique blend of the purest flavonoids, based on rigorous scientific research by Dr. Theoharides and his colleagues.
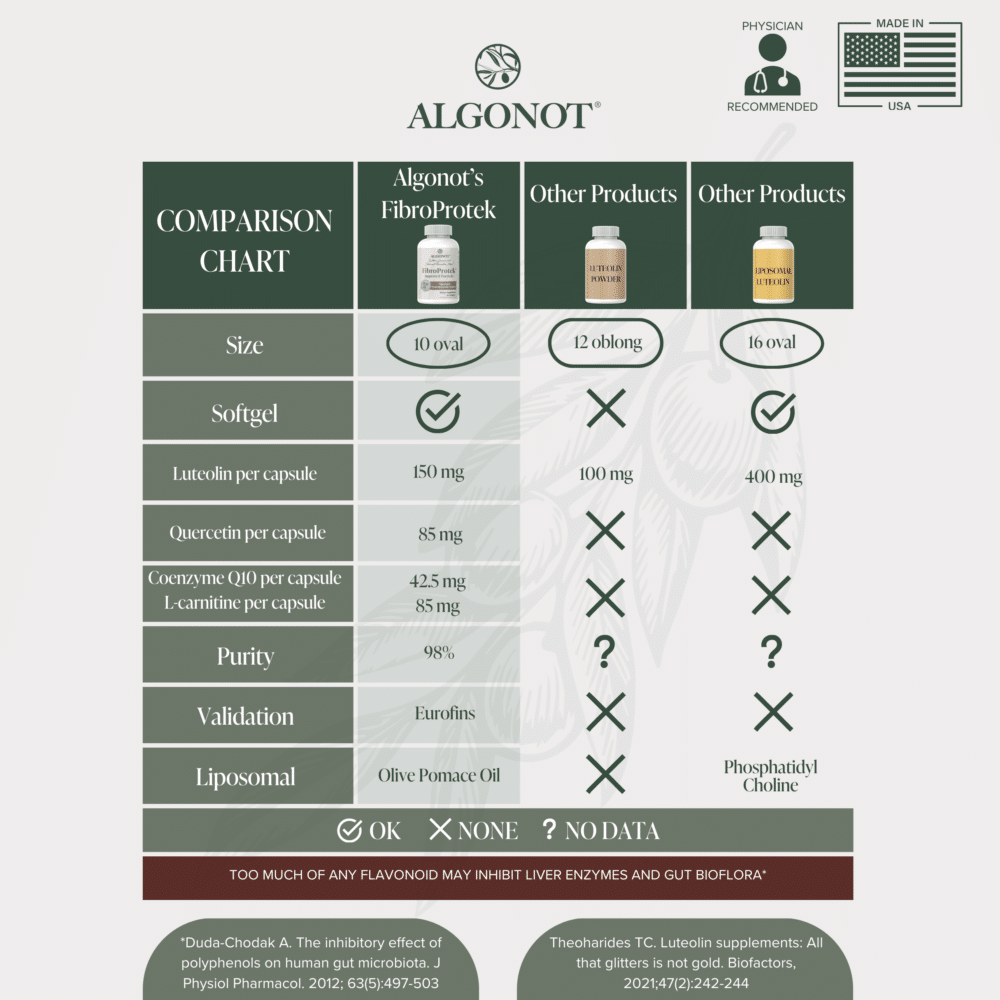
FibroProtek® is the only liposomal dietary supplement formulated using olive pomace oil, enhancing absorption and providing the well-known benefits of Mediterranean olives.
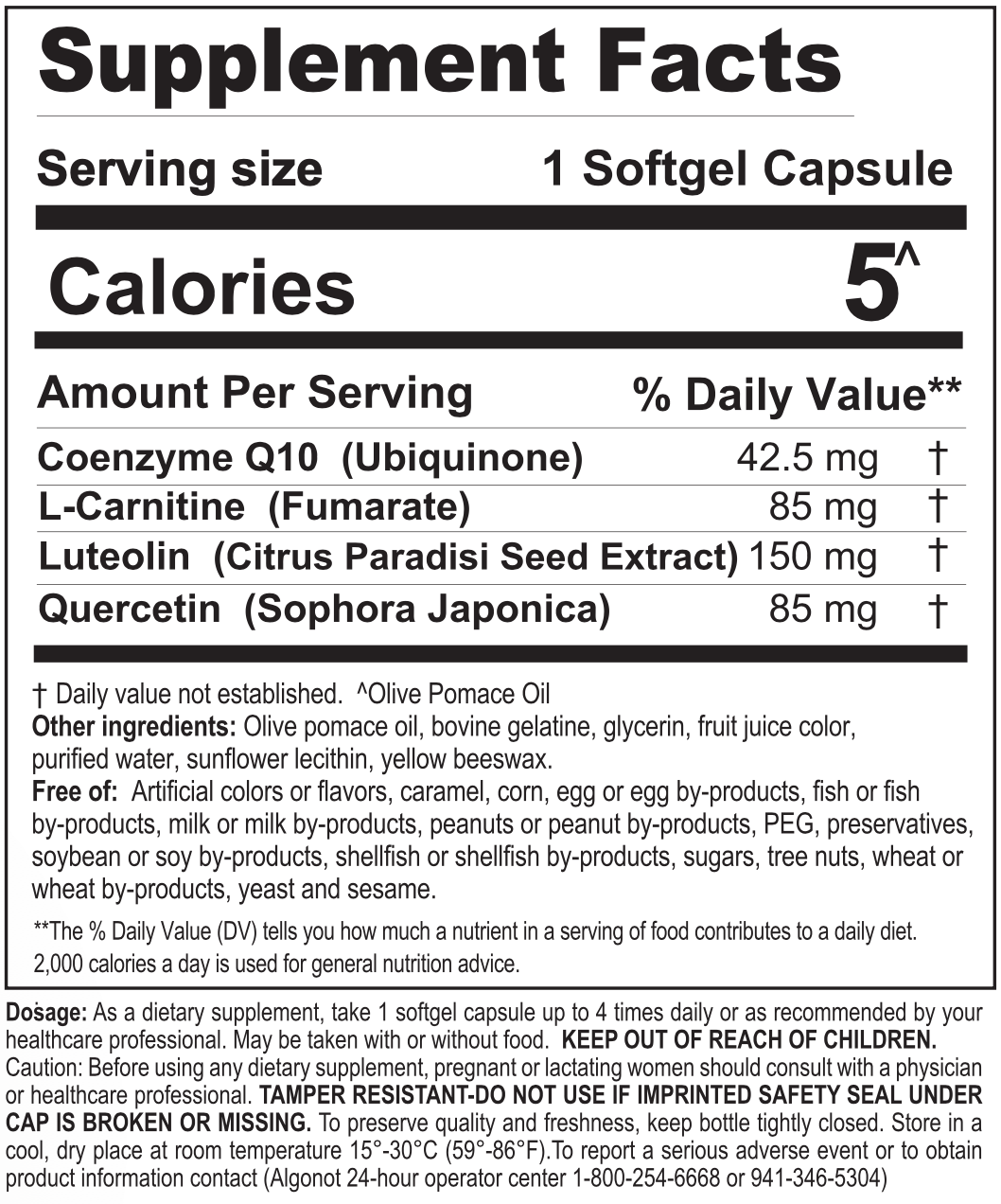
FibroProtek® is FREE of artificial colors or dyes, flavors or sweeteners, casein, corn, egg or egg by-products, fish or fish by-products, gluten, heavy metals, milk or milk by-products, peanut or peanut by-products, preservatives, glyphosate, salt, shellfish or shellfish by-products, soybeans or soy by-products, starch, tree nuts, wheat or wheat by-products and yeast.
Coenzyme Q10: helps convert food to energy and may be important to cognitive and nervous system health.
Carnitine: in studies, it’s been shown to ease discomfort, lessen fatigue and improve mental health.
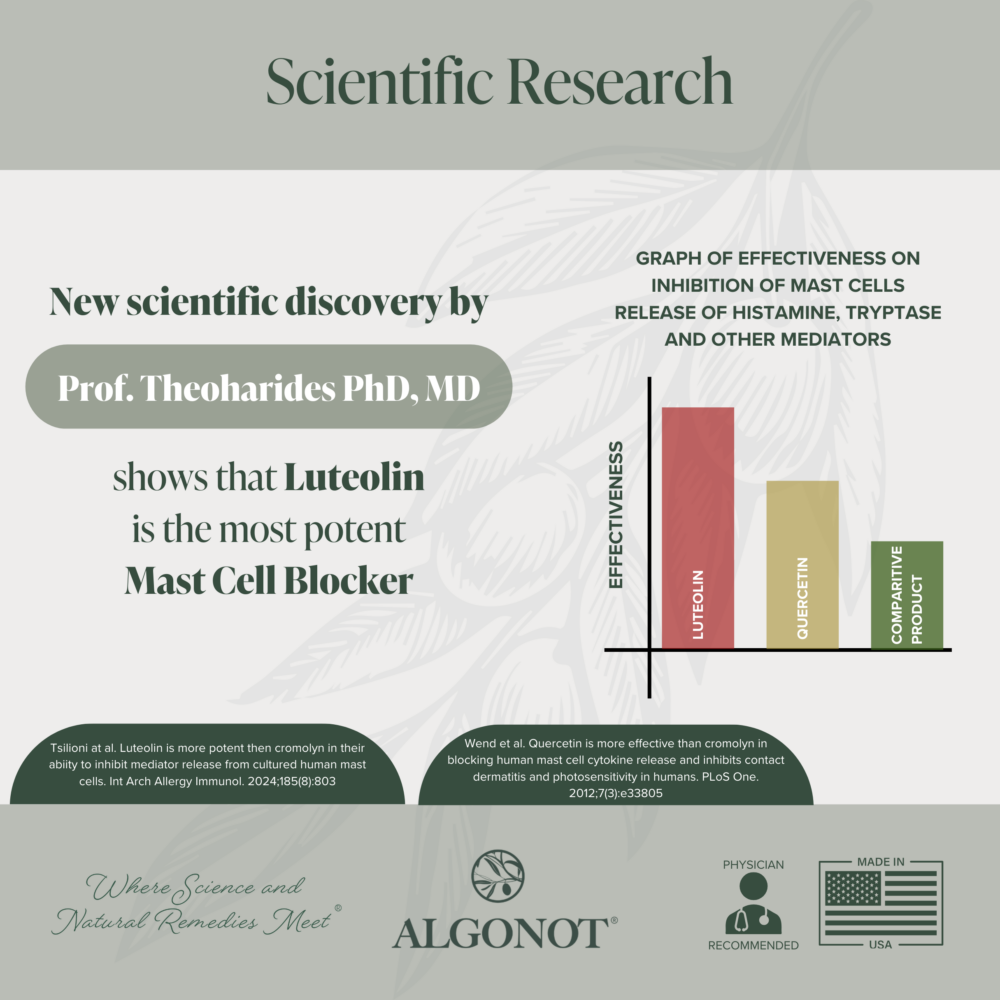
Luteolin: an antioxidant that may reduce free radicals, mast cell and microglial activation, according to recently published research.
Quercetin: a natural non-acidic flavonoid, obtained from the sophora plant.
Olive Pomace Oil (Unprocessed, Microfiltered): Unique to the Algonot® formulations is olive pomace oil, which is instrumental in increasing the absorption of flavonoids. Contrary to competitors who use seed oils, pomace oil is unsaturated, providing the well-known benefits of Mediterranean olives.
The flavonoids in FibroProtek® and other Algonot® products are derived from citrus seed extract and the sophora japonica plant, unlike competitors who commonly use fava beans or peanut shells that may lead to adverse reactions in individuals with sensitivities.
Manufacturing: Algonot® diligently oversees its vital manufacturing process to guarantee quality at each phase. All ingredients and finished dietary supplements undergo third-party testing from Eurofins to verify their purity (98% pharmaceutical grade) and adherence to truth in labeling standards. Algonot® dietary supplements are produced according to our precise specifications at Tishcon Corporation, a cGMP-certified facility in the United States, and are certified by NSF International, an esteemed organization that validates compliance with industry standards. This guarantees that every aspect of production is thoroughly examined and meets all technical and FDA regulations.